ISO 9001 Procedures Manual
Create an ISO 9001 Procedures Manual fast. Save time writing out quality procedures. Our Quality Management System (QMS) comes with the basic components to get your started. The process that your organization must show that it can comply with regulatory requirements and implement the system successfully in order to be truly helpful to your clients in order to receive ISO 9001 certification.
Are you implementing an ISO quality program at your organization? What will be your first step? How will you get started? Our ISO 9001:2015 Procedure Templates will answer many of these questions.
“We are a small credit counseling business in the Midwest. Our industry requires ISO certification. We decided to purchase the ISO 9001 Manual even though our industry does not require the same procedures as a manufacturer. I was impressed with the details of their manual and was able to extract sufficient information to complete our ISO 9001 certification in nine months. I would recommend the manual to any manufacturer seeking certification. All the information contained in the manual may not be pertinent to your business but should at least guide you in the proper components required.”
Robert Kory
Falls Credit Management, Inc.
ISO 9001 Quality Procedures
We have heard from several customers about the need for implementing ISO 9001 quality procedures in their unique organizational settings. If you are asking ‘How do I get started?’ or ‘How do I roll out ISO 9001:2015 in my company?’ Then you have come to the right place.
Implementing the ISO 9001 Quality Management System can serve as a business improvement tool. The ISO 9001 standard is perceived as a checklist item; Get ISO 9001 Certified-Done. The real goal of ISO 9001:2015, however, is continual improvement of the organization. Embracing the Management System Standards of ISO 9001 can enhance an organization.

ISO 9001 Certification Procedures
Introducing our comprehensive ISO 9001 Procedures Manual, a meticulously crafted and time-tested solution designed to streamline your organization’s quality management processes and guide you towards ISO 9001 certification with ease.
Key ISO 9001 Manual Features
We understand the importance of maintaining consistent quality standards to meet customer expectations and stay competitive in today’s market. Our ISO 9001 Procedures Manual is thoughtfully developed by industry experts, keeping in mind the diverse needs of businesses across various sectors.
Compliant with ISO 9001
Our ISO Procedures Manual aligns perfectly with the ISO 9001 standard, ensuring adherence to internationally recognized quality management practices.
Comprehensive and User-Friendly
With a clear and concise structure, our manual guides you through all the essential processes and requirements for ISO 9001 compliance. It is designed for easy navigation and understanding, making it accessible to all members of your organization.
Customizable ISO Templates
We provide fully customizable ISO templates that can be tailored to suit your specific business processes and organizational needs. This flexibility allows you to integrate our manual seamlessly into your existing quality management system.
Step-by-Step ISO Implementation
Our manual offers a step-by-step ISO implementation guide, empowering you to establish a robust Quality Management System (QMS) efficiently. It includes comprehensive procedures, forms, and checklists to streamline your QMS documentation.
Risk-Based Approach
Our manual emphasizes a risk-based approach to quality management, enabling you to identify, assess, and mitigate potential risks effectively, ultimately leading to improved operational efficiency and customer satisfaction.
Continuous Improvement
We encourage a culture of continuous improvement through our manual, guiding you to monitor, measure, and analyze performance data, helping you identify areas for enhancement and implement corrective actions.
Regulatory Compliance
Our manual assists you in meeting regulatory requirements and enables a smooth transition during external audits and certification assessments.
Whether you are starting from scratch or seeking to enhance your existing QMS, our ISO 9001 Procedures Manual is your ideal companion on the journey to operational excellence and ISO 9001 certification. Empower your team with the tools they need to achieve lasting success and customer satisfaction.
Don’t miss this opportunity to streamline your organization’s quality management practices and take your business to new heights of success. Invest in our ISO 9001 Procedures Manual today and unlock the full potential of your organization’s quality performance. Let’s embark on this transformative journey together.
Putting Together an ISO Quality Management System
We have put together what you will need to build a Quality Management System. It starts with a situational analysis to determine your interested parties, their wants, needs, opportunities and risks.
Customer Satisfaction
Customer Satisfaction is the goal of ISO companies. Customers, owners, suppliers, employees, the community and others that have a stake in the ongoing success of your organization. Top Management should use the results of the situational analysis in the development of the Company’s QMS Scope, Quality Policy, Quality Objectives, and QMS processes to enhance customer satisfaction.
QMS Scope
QMS Scope, consists of defining the requirements of relevant interested parties, the types of products and services covered by the QMS, and provide justification for any exclusions to requirements of ISO 9001 that the organization determines is not applicable to the scope of its quality management system.
Note: Exclusions to ISO 9001 may only be claimed if they do not affect the organizations ability or responsibility to ensure the conformity of its products and services and the enhancement of customer satisfaction.
Quality Policy
Quality Policy, must be appropriate to the purpose and context of the company. It ties in with and provides a framework for the Quality Objectives, includes a commitment to satisfy requirements, and a commitment to continual improvement of the QMS.
Quality Objectives
Quality Objectives, are derived from the Quality Policy, are measurable, and communicate to interested parties the effectiveness of the QMS. As quality objectives, they provide a basis for planning that at a minimum includes: Who, will do What, by When, requiring What resources, and How will results be evaluated.
QMS processes
QMS processes are those necessary for the QMS. Each QMS process shall identify:
• the inputs and outputs expected,
• the sequence and interaction of these processes with each other,
• measurable process criteria (or control method),
• resources required by the process,
• responsibility for the process,
• process risks (and approach to controlling risk),
• how the process will be evaluated, and
• opportunities for improvement.
ISO Quality Manual
Next, you can can create a Quality Manual. A Quality Manual explains the “scope” of your system. It is not required anymore, but it helps to explain what the boundaries are (scope), your Quality Policy, Quality Objectives, and QMS Processes.
We provide two versions of the Quality Manual. A shorter “lean” version and a longer version. The short version of the Quality Manual reflects lean thinking most of all. Rather than borrow the text of the standard verbatim, we stripped the Quality Manual to the bare essentials.
It’s not going to be for everyone – some will, no doubt, still insist that their ISO Quality Manuals parrot the text of the ISO 9001 standard, so that they’re sure their Quality Management System documentation covers every aspect of ISO 9001.
Putting in a lot of verbiage that’s not required by the standard, however, it can make your Quality Manual less user-friendly, less useful, and less likely to be improved. In fact, the new ISO 9001:2015 does not require a Quality Manual at all. But just in case this is your approach, we also provide a long version of the quality manual that covers all of the text of the ISO 9001 standard — all 123 shall statements.
There is a new term “Documented Information” that has replaced required procedures. There are 24 quality procedures in the new and improved ISO 9001:2015 QMS. Quality procedures that are included are ones that many organizations consider important or critical (e.g., Internal Auditing, Corrective Action).
Those ISO procedures are modeled on the Deming Cycle (PDCA) — they have a Plan, Do, Check, and Act phase, also known as the Process Approach. Some procedures that aren’t typically high priority and/or that didn’t amount to much more than simple work instructions (e.g., Serial Number Designation) have been omitted.
ISO 9001 Control of Documented Information (clause 7.5.3)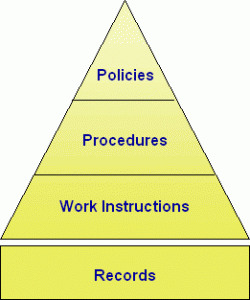
Let’s address what we feel is a real misconception about ISO 9001; the documentation requirements. It’s gone! First; the ISO 9001 standard does not have complex or complicated documentation requirements. In fact, they are quite simple; ISO 9001 Management System Standards no longer requires specific procedures. What does this mean?
You specify what you need. If your need work instructions then create them. If you workforce is quite skilled and your product is does not vary a lot, then maybe you just need training material. It’s your decision.
While it may difficult for most organizations to get by with no procedures, the fact that there are no ISO required standard operating procedures should send a message that, despite the perception of the opposite, ISO 9001 is not about a lot of procedures. Your organization must decide what procedures are needed based on issues related to compliance, importance, and performance.
An important concept to understand is that procedures are only one way to document processes, and the ISO standard recognizes that. Processes can be documented by Work Instructions, Visuals Aids, or training materials. The real requirement by ISO is that key business processes are understood and consistently carried out.
Throughout the updated manual, we refer to ISO 9001 requirements. We’ve done something new with the “Forms and Records” section at the end of each procedure. We’re still providing sample forms for you to use, but now they’re categorized with the iso 9001 version 2015 clause that requires the record. For example, in the Internal Audits procedure, there’s a form for audit reports; the Forms/Records table tells you the report requirement is in clause 9.2.2.
We think this is a much improved product, one that will smooth the bumps in your organization’s road to ISO 9001:2015 compliance and certification. Download Free Sample ISO Procedures to see how easy it is to edit MS Word Templates to build your own policy and procedure quality management system.
Fast ISO 9001 Procedures Manual
The ISO 9001 Procedure Manual is written from a manufacturing perspective, yet the quality policies and procedures can be generalized for any business. An ISO 9001 Quality Procedures Manual simplifies the task of complying with the ISO 9001 standard.
With the purchase of this ISO 9001 Procedure Manual, you get 24 ISO procedures, 57 quality forms/records, 25 job descriptions, background on ISO 9000, a two sample ISO Quality Manual, and guidelines to help you develop an effective quality management system (QMS).
What’s Included in the ISO 9001 Procedures Manual?
You will receive Quality Manual, 24 procedures, 57 forms, and 25 job descriptions pre-written in Microsoft Word for easy editing. You will receive 536 pages of content within seven sections:
1. ISO 9001 Quality Manual Template
First, two choices: a 20 page (long) or 4 page (lean) Quality Manual is used as a top level Quality document governing your company’s Quality Procedures.
2. ISO Manual Table of Contents
Next, 302 pages that include 24 prewritten ISO Quality Procedures, 57 ISO forms and corresponding activities–customize each Microsoft® Word document template to your needs.
3. ISO Job Descriptions
The ISO 9001 Manual also includes 80 pages with 25 ISO Job Descriptions covering the primary ISO Quality Functions.
4. ISO 9001 Introduction Training Guide
A 60 page training guide explains the basic concepts of ISO Quality Management (QMS), its structure, standards and definitions, and more.
5. “How To” ISO Manual Preparation Guide
A 38 page Procedure writing guide provides guidance to develop and implement your own ISO Quality Procedures.
6. ISO 9001 Manual Keyword Index
Easily find what you are looking for inside the ISO 9001 Manual using the 26 page Keyword Index.
Fast ISO 9001 2015 Quality Procedure Templates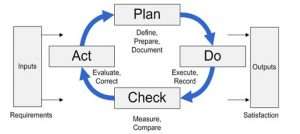
The Bizmanualz ISO 9001 Procedures Manual comes with over 536 pages of quality procedures and work instructions documentation (download only). All policies and procedures contained in the ISO 9001 manual are modeled on “Plan-Do-Check-Act” or PDCA Cycle.
You also get an explanation of the ISO 9001 2015 standard and helpful guidance to help you produce your company’s ISO 9001 procedures manual.
Free Sample ISO 9001 2015 Quality Procedure
Collect your Free ISO 9001 2015 Procedures. This sample Word files provides an example of the look and feel form the ISO Manual. Your will see how the header block is structured, revisions section, the style of write, content and more.
Take advantage of this comprehensive ISO 9001 Procedures Manual. Get your ISO 9001 certification journey started the right way — use the ISO 9001 QMS manual to simplify the process of developing, implementing, and managing your quality management system effectively! Download Free ISO 9001 2015 procedures.


















Frederick Owusu-Gyamera (verified owner) –
Very helpful
Suzie Adrina –
Good product and easy to use
Dane H –
It is a brilliant product! Thank you
Alan –
A useful starting point for production of manual and procedures. Somewhat expensive for what is generic material spread across a number of disciplines.
Overall Impression: Good
Quality Manager –
Documents we ordered were easy to find and to use and were available immediately.
Derick Pogieter –
Using the provided documents as acquired from Bizmanualz is a real pleasure. Documentation was written in a manner that is understandable, and user friendly by all my staff. Having the procedures in place, it was quite easy to adopt, and adapt to what we required in our company.
The lay-out of the manual is commendable – it is easy to reference and look-up.
The sample documentation was thoroughly thought through.
William Norwood –
We saved a lot of time and work and ended up with a 1st class set of manuals.