ISO 9001:2015 Introduction
An introduction to the ISO 9001;2015 standard explains ISO 9001 certification and how to get certified, and tells how your company will benefit from implementing and continually improving an ISO 9001 quality management system. The ISO 9001 Introduction covers the following:
ISO 9000 BACKGROUND
- Understanding the Goal of Implementing ISO 9001
- ISO Background
- From 9000 to 9001: An Evolution
- The Update to ISO 9001:2015
QMS Documentation
Quality Manual Contents
Quality Policy vs. Objectives
Quality Management System vs. Management System
Risk-Based Thinking vs. Preventive Action
Tools used for risk assessment
ISO 9001:2015 OVERVIEW
- Clauses 1-3 – General Information
- Clause 4 – Context of the Organization
- Clause 5 – Leadership
- Clause 6 – Planning
- Clause 7 – Support
- Clause 8 – Operations
- Clause 9 – Performance Evaluation
- Clause 10 – Improvement
BENEFITS OF USING ISO 9001:2015
- Employing ISO 9001 Improves Competitiveness
What is the Goal, Improvement or Certification?
Using the Process Approach
The Most Important Person in Your Business: The Customer
Supplier Relationships Enhance Effectiveness - Employing ISO 9001 Improves Organizational Culture
Fact-Based Decision Making
Establishing a Known Direction
Participative Management - Employing ISO 9001 Improves Documentation
Avoiding the ISO 9001 “Documentation Nightmare”
The Importance of Data Documents
BECOMING ISO 9001:2015 CERTIFIED
- Understanding ISO 9001 Certification
- Starting the Journey to ISO 9001 Certification
- Phase 2 – the Next Step in the ISO 9001 Journey
- Phase Three of Building a QMS System – Management and Resources
- Final Steps in Achieving ISO 9001 Certification
IMPROVEMENT THROUGH ISO 9001 MATURITY
- Understanding ISO 9001 Certification
ISO Organization Has No Role in Gaining Certification
The International Accreditation Forum
The Importance of Selecting a Registrar
Other Factors to Consider When Selecting a Registrar
Starting the Journey to ISO 9001 Certification
Create an ISO Implementation Plan - Phase 1 – Start with Clause 4 of the ISO 9001 Standard
Define Your ISO Processes
Creating the First Level of ISO 9001 Documents
Completing Phase One with a Quality Policy, Objectives, and Quality Manual - Phase 2 – the Next Step in the ISO 9001 Journey
Sketch Out the Continual Improvement Processes
Drafting Continual Improvement Documents
Addressing Risk Based Thinking
Additional ISO System Components of Continual Improvement - Phase 3 – Management and Resources
Management Commitment is Vital to ISO 9001 Success
ISO 9001 Application Depends on the Organization
Focus on ISO Compliance – Not Unnecessary ISO Documentation
Training Documents Are Important in ISO 9001 - Final Steps in Achieving ISO 9001 Certification
Get a Handle on Your ISO 9001 Data Documents
Understand Specific ISO 9001 Requirements for Documents
Using ISO 9001 Records for Continual Improvement
Employ the ISO 9001 Process Approach
Try to Simplify ISO Compliance
Using and Auditing ISO 9001 QMS
IMPROVEMENT THROUGH ISO 9001 MATURITY
- Phase One – Realizing the Need for Process Improvement
Using the Maturity Model to Kick Start Your Process Improvement
- Phase Two – Collecting Management System Data
Policies, Procedures and Forms are Key Documentation
- Phase Three – Separating the Common from the Special
Using Data Analysis to Achieve Stability
- Phase Four – Finding Root Causes
Achieving Stability Allows You to Focus on Regularly Occurring Problems
- Phase Five – Risk Management
Implementing Effective Management Systems
USING YOUR ISO 9001 QMS MANUAL AND ACCREDITATION
ISO DEFINITIONS
Terms and definitions for use with building an ISO 9001 registered quality management system.
ACCREDITATION
The act of giving an organization formal approval to operate an audit and registration program.
ACCREDITATION BODIES
IAF member countries have organizations that are chartered to accredited Registrars. In the USA, it’s the ANSI-ASQ National Accreditation Board (ANAB); in Canada, the Standards Council of Canada (SCC); in the Netherlands, the Raad voor Accreditatie (RvA). These ISO Accreditation Bodies publish the requirements that they set forth for Registrars to become accredited. These requirements generally follow other ISO documents. The Accreditation bodies regularly audit registrars’ procedures, systems and audit practices to ensure they meet and maintain systems to the requirements.
AMERICAN SOCIETY FOR QUALITY (ASQ)
The ASQ established the Registrar Accreditation Board (RAB) in 1989. In 1991, ANSI and RAB joined forces to establish the American National Accreditation Program (NAP) for Registrars of Quality Systems. On January 1, 2005, RAB and the ANSI-RAB NAP was replaced by the ANSI-ASQ National Accreditation Board (ANAB).
AUDIT STANDARD
An authentic description of essential characteristics of audits, which reflects current thought and practice.
AUDITEE
The organization being audited to an ISO Standard.
AUDITORS
ISO Auditors work for or contract to Registrars to perform registration assessments and surveillance They are the “front line” in the process. Registrars are responsible for ensuring Auditors meet qualification requirements. Their requirements include training in auditing, ISO 9001 training, and at least one member of the audit team must have experience in the industrial sector of the company being audited. Auditors collect the objective evidence demonstrating the effectiveness (or lack thereof) of the company’s quality management system and make registration recommendations to the Registrar. The Registrar has the ultimate decision however.
ANSI
The American National Standards Institute, a private, non-profit organization that administers and coordinates the U.S. voluntary standardization and conformity assessment system.
AUDITING ORGANIZATION
A unit or function that carries out audits through its employees. This organization may be a department of the auditee, client or an independent third party. See “Auditors”.
CAPABILITY
Ability to perform designated activities and to achieve results, which fulfill specified requirements.
CERTIFICATION
The authoritative act of documenting compliance with agreed requirements. See Top 10 Reasons Why You Need ISO 9001 Certification.
CERTIFICATION BODY
An impartial organization possessing the necessary competence to operate a certification program.
CHARACTERISTIC
A physical, chemical, visual functional or any other identifiable property of a product or part or material.
CLIENT
The person or organization requesting the audit. Depending on circumstances, the client may be the auditing organization, the auditee or a third party.
COMPANY
Term used primarily to refer to a business first party, the purpose of which is to supply a product or service.
COMPLIANCE
A judgment that a product or service meets or “conforms” to the requirements of a specific standard. See ISO Quality Standards.
CONCESSION
Formal act of yielding, waiving, or surrendering a right or privilege. A concession is often made in writing by a customer when it has been determined that a product or contractual requirement cannot be met in a practical or cost-efficient way and that dropping the requirement will not materially affect the desired outcome (or may even improve it). Also see “waiver”.
CONFORMITY
The fulfillment of specified requirements. Same as compliance.
CONTRACTOR
The supplier.
CONTRACT REVIEW
Critical evaluation of a binding agreement (contract) between parties, for the purpose of ensuring that requirements are clearly and adequately stated so that each party understands the requirements placed on it by the contract, as well as ensuring that each party is capable of meeting – and is willing to meet – its contractual requirements. A contract review is done before the contract is made formal and legally binding (by the signature of both parties, for instance).
CORRECTION
Actions taken to correct or eliminate a detected nonconformance, defect, or problem.
CORRECTIVE ACTION
A common ISO Definitions term, it represents a process, or series of actions, designed to minimize or eliminate the likelihood that a nonconformance, defect, or problem will recur, or happen again; not to be confused with “correction”. See Understanding Corrective Actions.
CRITICALITY
A relative measure of the consequences of a failure mode and its frequency of occurrence.
CUSTOMER
Ultimate consumer, user, client, beneficiary or second party.
DEFECT
The non-fulfillment of intended usage requirements. The departure or absence of one or more quality characteristics from intended usage requirements. See nonconformance.
DEGREE OF DEMONSTRATION
The extent to which evidence is produced to provide confidence that specified requirements are fulfilled.
DEPENDABILITY
Capability of meeting a specified set of requirements, usually established by repeated use over an extended period of time; trustworthiness; reliability.
DESIGN REVIEW
A formal, comprehensive examination of a product design and the product design phase, where design capabilities are compared with design specifications (requirements), problems are identifed, and solutions are proposed. Design reviews may be conducted at any point in the design process.
DESIGN SPECIFICATIONS
A description of the physical and functional requirements for a product. In its initial form, the design specification is a statement of functional requirements with only general coverage of physical and test requirements. The design specification evolves through the research & development phase to reflect progressive refinements in performance, design, configuration and test requirements.
DESIGN TRANSFER
The transfer of the design basis or baseline into specifications for the product, its components, packaging, labeling, and the manufacturing and quality assurance procedures, methods, specifications, etc., so that the product can be produced using production methods.
DISCREPANCY
A failure to meet the specified requirement, supported by evidence (Also can be called Nonconformance, Deficiency or Finding).
DISPOSITION
Resolution (as of a matter); settlement. For example, “The final disposition of the returned material is determined by the Quality Manager”.
ISO DOCUMENT
Something written or printed that provides factual information or proof; any object used as evidence; to prove or support by means of documents.
ENVIRONMENT
The conditions, circumstances, influences and stresses surrounding and affecting the product during manufacturing, storage, handling, transportation, installation and use.
FAILURE
An event in which a previously acceptable product does not perform one or more of its required functions within the specified limits under specified conditions.
FAILURE ANALYSIS
The logical, systematic examination of an item, including its diagrams or formulas, to identify and analyze the probability, causes and consequences of potential and real failures. See Failure Modes Effects Analysis (FMEA).
FAILURE CAUSE
The physical or chemical process, design defect, quality defect, component misapplication, or other processes which are the basic reason for failure or which initiate the physical process by which deterioration proceeds to failure.
FAILURE EFFECT
The consequences a failure has on the operation, function, or status of a product.
FAILURE MODE
The manner in which a failure is observed. The way a failure occurs and its impact on the product performance.
FAILURE MODE AND EFFECT ANALYSIS (FMEA)
The process of identifying potential weaknesses or risks. A design FMEA reviews schematics, engineering drawings, etc., to identify basic faults at the part/material level and determine their effect at finished or sub – assembly level on safety and effectiveness. A process FMEA is the same but looks at each activity or task within a process to determine risks and possible actions or controls to reduce risks.
FAILURE PATTERN
The occurrence of two or more failures of the same component or feature in identical or equivalent application, which are caused by the same basic failure mechanism.
FAULT TREE ANALYSIS
The process of identifying potential design weaknesses using a highly detailed logic diagram depicting basic faults and events that can lead to system failure and/or safety hazard.
IAF
The International Accreditation Forum, the world association of conformity assessment accreditation and other bodies interested in conformity assessment in the fields of management systems, products, services, personnel and other similar conformity assessment programs. Its primary function (according to the “About IAF” page) is to develop a single worldwide program of conformity assessment which reduces risk for business and its customers by assuring them that accredited certificates may be relied upon.
ISO
ISO comes from the Greek word ‘isos’ for equal. It also refers to International Organization for Standardization, a network of hundreds of national standards institutes that is, according to its web site, the world’s largest developer and publisher of international standards. Its Central Secretariat. See What Does “ISO” Mean? and What is ISO?
ISO MEMBER BODY
An organization that is chartered by ISO to accredit Registrars in a member country. “Member body” is actually the highest of three ISO member levels; the others are correspondent members and subscriber members. A member body can take part in any ISO technical or policy committee and cast votes.
LEAD AUDITOR
The individual appointed by the registration organization to be responsible for the quality audit and the audit team is the Lead Auditor.
MANAGEMENT REVIEW
A formal examination of a project and its status, successes, problems encountered, etc., compared with project objectives. The management review is conducted by “top management”. which usually consists of a chief executive officer, a chief finance officer, the ISO management Representative, and the quality manager, at a minimum.
NATIONAL ACCREDITATION PROGRAM
In 1991, ANSI and RAB joined forces to establish the American National Accreditation Program (NAP) for Registrars of Quality Systems. On January 1, 2005, RAB and the ANSI-RAB NAP was replaced by the ANSI-ASQ National Accreditation Board (ANAB).
NONCONFORMANCE
A condition of any product or component in which one or more of its characteristics do not conform to requirements. Nonconformances include product (part) failures, deficiencies, defects, and malfunctions. A nonconformance may also be referred to as a “nonconformity”.
NIST
National Institute of Standards and Technology; a non-regulatory federal agency within the U.S. Commerce Department’s Technology Administration. Its stated mission is to develop and promote measurement, standards, and technology to enhance productivity, facilitate trade, and improve the quality of life.
OBSERVATION
A statement of fact made in an audit team’s report that something was found during the audit that doesn’t rise to the level of a nonconformity (no objective evidence of nonconformity, doesn’t require a corrective action) but which, if left alone, could result in a future audit finding. Sometimes referred to as an “opportunity for improvement”.
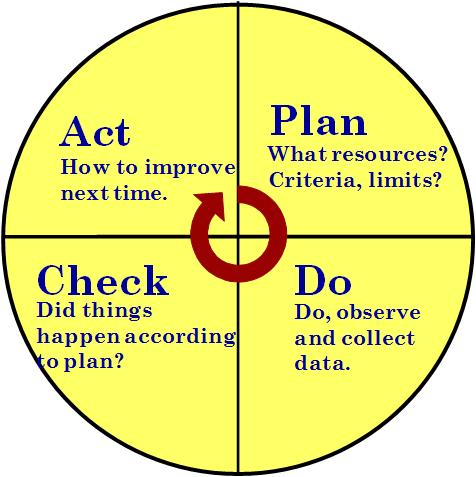 PLAN-DO-CHECK-ACT
PLAN-DO-CHECK-ACT
The PDCA process-based improvement cycle used in both ISO 14000 and ISO 9001 standards. See How Are PDCA Cycles Used Inside ISO 9001?
POLICY
A definite course or method of action to guide and determine present and future decisions. It is a guide to decision making under a given set of circumstances within the framework of corporate objectives, goals and management philosophies. ISO mandates a Quality Policy be developed and used as a basis for your quality objectives.
PREVENTIVE ACTION
An action taken to eliminate the causes of a potential nonconformity, defect or other undesirable situation, to prevent occurrence. See Understanding Preventive Actions.
PROCEDURE
A particular way of accomplishing something, an established way of doing things, a series of steps followed in a definite regular order. It ensures the consistent and repetitive approach to actions. Quality Procedures are procedures that meet or exceed ISO 9001 requirements.
PROCESS
A set of interrelated resources and activities that transform inputs into outputs with the aim of adding value. Resources include personnel, facilities, equipment, technology, methodology and finances. The aim of adding value if quality related.
PRODUCT
The result of activities or processes. A product can be tangible or intangible, or a combination of both.
PRODUCTION PERMIT
A written authorization for a product, before its production, to depart from originally specified requirements; also known as deviation.
PURCHASER
The customer.
QUALIFICATION
A documented determination that a product (and possibly its associated software, component, packaging, or labeling) meets prescribed design and performance requirements.
QUALITY
The composite of all the characteristics, including performance, of an item, product or service that bear on its ability to satisfy stated or implied needs. In a contractual environment, needs are specified, whereas, in other environments, implied needs should be identified and defined. In many instances, needs can change with time; this implies periodic revision of requirements for quality. Needs are usually translated into characteristics with specified criteria. Quality is sometimes referred to as “fitness for use”, “customer satisfaction”, or “conformance to the requirements.”
QUALITY ASSURANCE
A planned and systematic pattern of all actions necessary to provide adequate confidence that the product, its components, packaging and labeling are acceptable for their intended use.
QUALITY AUDIT
Methodical examination of an organization’s quality management system, in whole or in part, to assure conformance to a specific quality standard (e.g., ISO 9001). Quality audits may be conducted by external (third) parties or they may be done internally. “Certification audits” must be conducted by accredited third-party auditors.
QUALITY AUDITOR
One who conducts a quality audit; must be qualified to conduct quality audits. Also see “lead auditor”.
QUALITY DOCUMENT
A document that contains requirements for quality system elements for products or services. The results of activities such as inspections or quality audits.
QUALITY IMPROVEMENT
The actions taken to increase the value to the customer by improving the effectiveness and efficiency of processes and activities throughout the organizational structure.
QUALITY MANAGEMENT PRINCIPLES
Used by management as a guide towards improving performance. The Quality Management Principles were derived from the experience of experts on the technical committees and represent the main elements that a good quality system must have. The eight principles are: Customer Focus, Leadership, Involvement of People, Process Approach, Systems Approach to Management, Continual Improvement, Factual Approach to Decision-making, and Mutually Beneficial Supplier Relationship.
QUALITY REQUIREMENTS
A translation of customer needs into a set of quantitatively or qualitatively stated requirements for the characteristics of a product or service to enable its realization and examination. The requirements for quality should be initially expressed in functional terms and documented.
RAB
Registrar Accreditation Board. The U.S. organization for accreditation of Environmental Management Systems ISO 14001, Quality Management Systems ISO 9000 registrars, and auditor training course providers. On January 1, 2005, RAB and the ANSI-RAB NAP was replaced by the ANSI-ASQ National Accreditation Board (ANAB).
REGISTRAR
Organizations that issue ISO certification. Also called “certification bodies” or “registration bodies”. Their place in the system is to verify companies meet the requirements of ISO 9001:2000 and certify or register companies that do. Registrars must meet the requirements of the Accreditation Bodies. These requirements include things such as independence; Registrars cannot consult for instance. This system ensures uniformity in the registration process.
REGISTRATION
A formal verification process by an accredited body that an organization has been audited and shown to comply with ISO 9000.
RELIABILITY
The characteristic of a product, or any component thereof, expressed as a probability that it would perform its required functions under defined conditions for specified operating periods.
RELIABILITY ASSESSMENT
A quantitative assessment of the reliability of a product, system or portion thereof. Such assessments usually employ mathematical modeling, directly applicable results of tests on the product, failure data, estimated reliability figures, and non-statistical engineering estimates.
RVA
The Dutch council for accreditation. It was established to supervise and monitor organizations which judge quality systems and/or examine an organization on the basis of European and international standards and provide accreditation.
SCC
Standards Council of Canada, the ISO Member body chartered to accredit Registrars in Canada.
SEVERITY
The consequences of a failure mode. Severity considers the worst potential consequences of a failure, determined by the degree of injury.
SERVICE
Work performed by a person or group for the benefit of another. ISO standards include “service” in their definition of the term “product”, as in “The computer repair service is the service company’s product.”
SPECIFICATION
Documented detailed requirements with which a product or service has to comply.
SUPPLIER
The organization that provides a product or service. Same as subcontractor.
SYSTEM
The principal functioning entities comprising the product, e.g. hardware, software. Also an organized and disciplined approach to accomplish a task, e.g., a failure reporting system. The Quality Management System (QMS) consists of the realization and support processes required to ensure the products meet customer requirements.
TESTING
The determination by technical or scientific means of the properties or elements of a product or its components, including functional operation, and involving the application of established scientific principles and procedures.
TRACEABILITY
The ability to trace the history, application or location of a product and, in some cases, service by means of recorded identifications. Traceability may refer to: a product, a calibration and its relationship to the measuring equipment and the national or international standards, primary standards, basic physical constants, properties or references materials. Traceability requirements should be specified for some stated period of history or to some point of origin.
VERIFICATION
Act or process of establishing (confirming) the accuracy or existence of something; in the quality field, verification is a systematic, objective, and documented process of confirming that a product or service conforms to various requirements (customer, regulatory, etc.).
VALIDATION
Act or process of establishing that the results not only meet requirements but also satisfy the intended purpose. Learn how to use procedure validation.
WAIVER
Voluntary surrender of one’s privilege(s); relinquishing, or not insisting on, a right or privilege. In quality, a waiver is generally a customer’s written authorization that the vendor to release its product, even though the product does not conform to one or more requirements; also known as a “concession”.







Leave a Reply