What’s the Difference Between Procedures and Work Instructions?
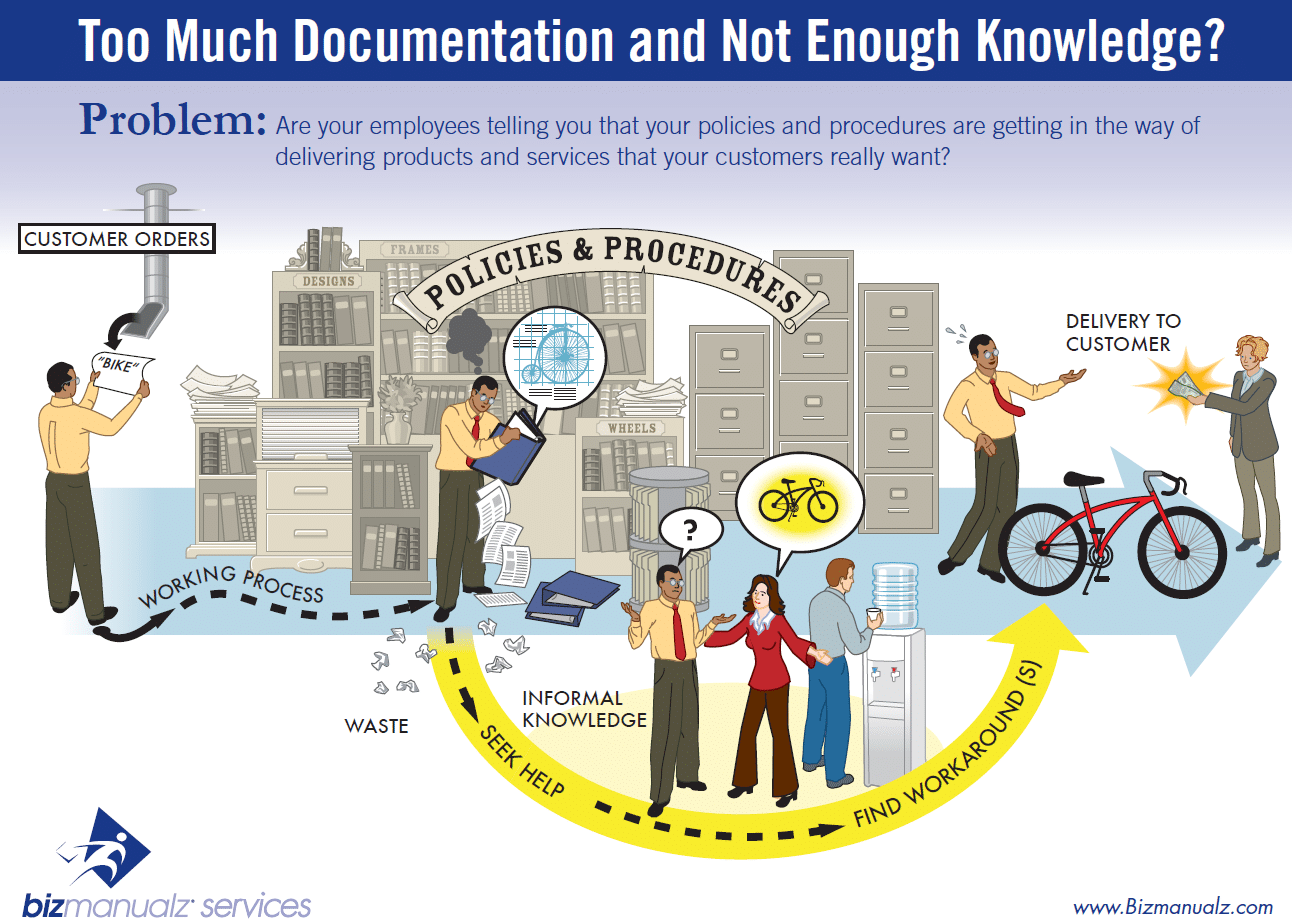
Many people confuse procedures and work instructions. In fact, most people write work instructions and call them procedures. Knowing the difference between procedures and work instructions can help you understand the documentation process much better and, therefore, develop great procedure documentation.
Procedures and Work Instructions
Procedures describe a process, while a work instruction describes how to perform the conversion itself. Process descriptions include details about the inputs, what conversion takes place (of inputs into outputs), the outputs, and the feedback necessary to ensure consistent results. The PDCA process approach (Plan, Do, Check, Act) is used to capture the relevant information. Questions that need to be answered in a procedure include:
- Where do the inputs come from (suppliers)?
- Where do the outputs go (customers)?
- Who performs what action when (responsibilities)?
- How do you know when you have done it right (effectiveness criteria)?
- What feedback should be captured (metrics)?
- How do we communicate results (charts, graphs and reports)?
- What laws (regulations) or standards apply (e.g., ISO 9001, 8th EU Directive, IFRS, Sarbanes-Oxley)?
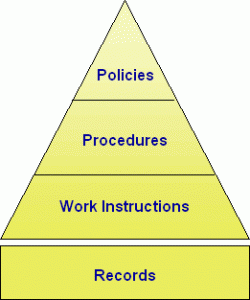
We can look at documentation as a pyramid comprised of four levels (or types of documents):
Level 1 and Level 2 represent your policies and procedures. What is the difference between policies and procedures? Your policies are at the top of the pyramid, like a beacon from a lighthouse. They direct the procedure in the proper direction and provide a foundation for the procedure’s description of the process being documented.
 Level 1 – Policies and Objectives
Level 1 – Policies and Objectives
A policy is used as a course of action to guide and influence decisions. In an ISO 9001 system this may be found in the “ISO 9001 2015 Procedures“. The purpose of this level of documentation is to state concisely the policies and objectives of the company, organization, or division. Policies are similar in some ways to mission statements.
At a minimum, the Quality Manual is required to describe the scope of your registrations, your exclusions from any ISO clauses, your quality processes, and your quality procedures.
Level 2 – Procedures
Your procedures are the second level of documentation, which should be more detailed and describe who does what and when (in sequence or order). Activities for a department or function might be listed along with the functional titles or positions responsible for the procedure. These procedures can be text based but many use a process map to communicate the information.
As you rewrite the procedures for your application, be sure to verify your modifications against the requirements of the ISO standard to ensure all requirements are still addressed. An example of where you may want to change the format is the Effectiveness Criteria section of certain procedures.
When determining these criteria, a holistic approach as to what is best for the company should be used. An obvious example involves Purchasing. In the past, Purchasing effectiveness was measured by the purchase price of materials. The obvious fallacy with this measure is that out of specification or marginal components result in increase costs downstream and may actually result in higher total costs.
Level 3 – Work Instructions
Work instructions or a Method of Procedure, should be very detailed on “how” to accomplish a specific job, task or assignment. For example, a work procedure could be developed for assembling the final housing of a product with step-by-step instructions including such detail as the torque requirements of the fastening screws.
Individual work instructions are very specific to an industry or company. Supplemental documentation may be used including User’s Manuals, Engineering or Technical Manuals, Technical Support notes, Manufacturing Notes, etc., in order to create detailed work instructions.
Level 4 – Forms and Other Documents
The last level of documentation includes forms used to create records, checklists, surveys, or other documents used in the creation of a product or service. Records are a critical output of any procedure or work instruction. They form the basis of your process communications, audit material, and process improvement initiatives.
Quality

The Bizmanualz Scoreboard displays key segment processes, clearly stated objectives, and on-going performance metrics.
With an ISO 9001 Quality Management System, you need to assure that a company or division will meet the ISO 9000 requirements and receive certification. The following process should take place:
- Management Decision and Commitment
- Adequate Training and Evaluation
- Compliance with Appropriate Standards
- Audit and Registration
ISO 9000 Quality Management Systems Implementation
Some of the obstacles that can interfere with successful ISO 9000 Quality Management System implementation and that must be avoided can include, unrealistic time frames, resistance to change, lack of management commitment, insufficient training, or subjective interpretation of the standards.
The areas most frequently resulting in Non-Certification by companies to date have been in document control, design control, purchasing, inspection and testing, quality systems, process control and inspection, or measuring and test equipment. Although all areas of the company’s quality assurance program are required to be in compliance with the standard, management should perform extra reviews to ascertain compliance in these above areas.
The appropriate personnel under the direction of management should review the standards and develop, implement and maintain a minimum set of quality systems and procedures to satisfy the ISO 9000 standard.
Further, these personnel will provide confidence to management that the intended quality is being achieved and is:
- Documented
- Demonstrable
- Effective
- Maintained
Learning how to write policies and procedures is easy once you understand the four levels of documentation and the difference between policies, procedures, and work instructions. Free policies and procedures from Bizmanualz make it even easier!


















I go through the explanation of on procedure and work instruction. I understand that procedure is outline of process and WI is details of that procedure. For example, a procedure for disposing a paper say:
*Dispose the paper in waste. bin”, while work instruction would be “tear the paper into small pieces of about 1 cm manually or by shredder and discrd the small pieces in waste paper bin”
Is it necessary to have two documents for one activity while this can be covered in one document.
I understand that procedure and work instructions can be used interchangeably and be adequately elaborated so that activity can be performed consistently in the same way.
You do not need two documents for simple processes. But if the process is more complicated, then two documents keeps the reader focused and will address difference readers concerns.
Those articles inspire me a lot for my business and organizational set up.