document
Microsoft Word is a powerful word processor, packing features such as line and page breaks. These breaks are important for adding structure to your document and creating a smooth flow. Let’s explore how to use them! How to use line and page breaks in Microsoft word.
Read moreSaving your work in Microsoft Word is a must-have skill and a critical feature of Word. Learn how to save & print in Microsoft word.
Read moreFormatting a Word document requires a few techniques and tools. These let users adjust the layout and appearance of their content. Options like changing margins, fonts, and adding headers and footers are all available in Word. How to format a document in word?
Read moreElevate your written work by utilizing Microsoft Word’s Table of Contents feature! Not only does it organize your ideas in a more aesthetically pleasing way, but it also saves both you and your audience valuable time. How to insert table of contents in Microsoft word?
Read moreDo you want to create an automated form in Word? It’s not as daunting as it seems! With the right knowledge and tools, you can do it like a breeze. This article will show you the step-by-step process without any coding or technical expertise. How to Create an Automated Form in Word.
Read moreTo effectively use citations in Microsoft Word, familiarize yourself with the importance of incorporating citations in your work. This Introduction section delves into the significance and benefits of using citations. Explore the sub-sections – the Importance of using citations in Microsoft Word – to better understand how citations enhance the credibility and integrity of your writing. […]
Read moreMicrosoft Word is a popular word processor with many add-ins that let you customize your experience. What add-ins are available for Microsoft word?
Read moreMicrosoft Word is a must-have for anyone wanting to quickly put text into their documents. Follow these simple steps and you’ll be a pro in no time! How to insert text in Microsoft word.
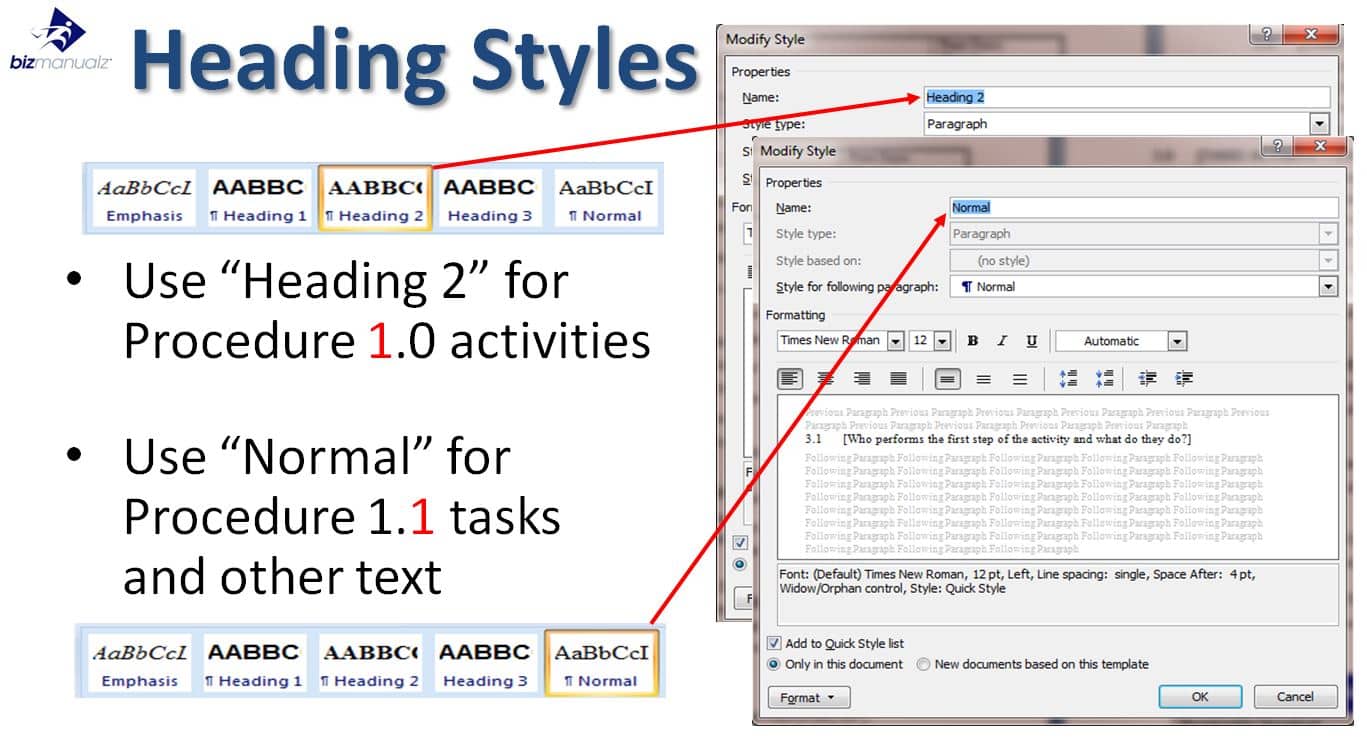
Read moreMicrosoft Word is a powerful tool with lots of features to help you format and style your documents. Styles are a great way to keep your docs looking professional and easy to read. Whether you’re writing a short letter or a complicated report, understanding how to use styles can save you time and keep your […]
Read more