What’s The Difference Between PRPs CCPs SSOP and GMPs?
Let’s start off with a blast! PRP’s, CCP’s, SSOP, and GMP’s – it may seem like alphabet soup at first. But don’t worry, we’ll unravel it all! It’s clear these acronyms play and important part in keeping consumers safe. The FDA even requires companies to use science-driven risk assessments to prevent food safety issues. So remember: without food safety regulations, it’s like playing Russian roulette with lunch!
Understanding Food Safety Regulations
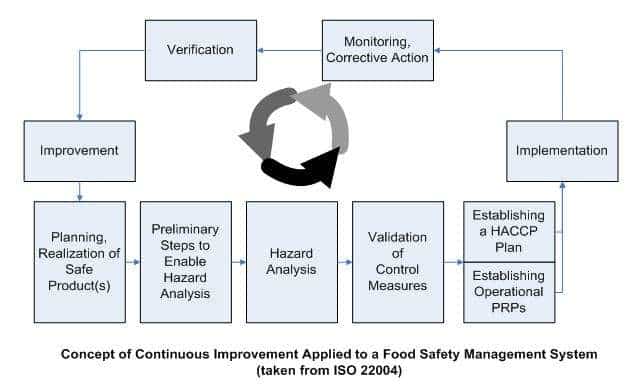
PRP’s stands for Prerequisite Programs. These are the foundation of food safety systems – like employee training and pest control measures. It’s a set of procedures and practices to create a hygienic environment. Employee hygiene training and pest control are examples.
CCP’s means Critical Control Points. These are special steps in production where hazards can be prevented or eliminated. These steps in the manufacturing process reduce hazards to and acceptable level. Monitoring these points helps identify and control hazards.
SSOP is Sanitation Standard Operating Procedures. They outline how to clean equipment and facilities to avoid cross-contamination. They provide written guidance on cleaning and sanitizing food processing equipment and facilities. It stops cross-contamination.
GMP’s means Good Manufacturing Practices. This is a guideline from regulatory agencies for manufacturers to follow during production processes. They involve guidelines and regulations to guarantee product quality and safety. GMPs cover personnel hygiene, facility maintenance, equipment calibration, and record keeping.
Local regulations should always be followed. Training for food handlers reinforces knowledge about these regulations and makes food production safer.
PRP’s – Prerequisite Programs
PRP’s, otherwise known as Prerequisite Programs, are key steps or processes that must be done before using more complex food safety systems. They form the basis for making sure food products are safe and of good quality.
To have a better understanding of PRP’s, let’s look into the different types:
| Type | Description |
|---|---|
| GMP’s (Good Manufacturing Practices) | These practices are essential in keeping a clean and hygienic manufacturing environment. This includes correct sanitation, pest control, staff training, and equipment maintenance. |
| CCP’s (Critical Control Points) | CCP’s concentrate on essential parts of the production process where preventive measures can be used to get rid of or minimize possible hazards. Monitoring and corrective actions play and important role in governing these points. |
| SSOP (Standard Sanitation Operating Procedures) | SSOP outlines step-by-step processes for powerful cleaning and sanitizing of machinery and facilities. Adhering to these procedures guarantees that potential contaminants are gotten rid of from the production environment. |
Implementing PRP’s is not only necessary for regulatory compliance but also for preserving customers’ health and building trust in your brand. By taking proactive measures to stop possible risks, businesses can protect their reputation, comply with food safety regulations, and in the end attain long-term success.
Start now! Don’t miss out on setting up strong PRP’s within your food production process. Prioritize food safety to reduce risks and guarantee the highest standard for your customers. Bear in mind, a secure foundation leads to successful growth!
 CCP’s – Critical Control Points
CCP’s – Critical Control Points
Let’s explore preventive measures of CCP’s.
Below is a table with info:
| Control Point | Description | Monitoring Method |
|---|---|---|
| Temperature | Ensure proper cooking/heating | Use calibrated thermometers |
| Time | Monitor duration of cooking/processing | Use timers |
| pH | Control acidity levels | Test using pH strips |
| Allergens | Prevent cross-contamination | Verify labels & segregation procedures |
| Packaging | Ensure proper sealing | Visual inspection & quality control checks |
These CCP’s are vital for product safety. We need more details. More than just monitoring, we need to set criteria for each point. Such as temperature ranges, timings, pH levels, segregation procedures for allergens, and packaging inspections.
By following these CCP’s, the risk of foodborne illnesses decreases. Plus, businesses meet regulatory requirements. So, take action now! Incorporate effective CCP’s in your production processes. To guarantee customer well-being and satisfaction. Do this with diligence, upholding the highest standards of food safety.
Why did the germ feel left out? Because it wasn’t invited to the SSOP party!
SSOP – Sanitation Standard Operating Procedures

MS Word Procedure Template
SSOPs, or Sanitation Standard Operating Procedures, are very important protocols. They ensure cleanliness and safety in many industries. Let’s explore them in detail!
To understand the importance of SSOPs, let’s see a table:
| Element | Description |
|---|---|
| Purpose | Outlining step-by-step cleaning procedures |
| Scope | Everything from equipment to personnel |
| Requirements | Guidelines for cleaning agents, frequencies, verification processes |
| Implementation | Training programs to teach proper sanitation practices |
| Monitoring | Inspections & audits to ensure compliance |
SSOPs involve personal hygiene and cleaning methods. They are part of Good Manufacturing Practices (GMPs). Their focus is on preventing cross-contamination and maintaining product quality.
Once, there was a food processing plant with no sanitation procedures. Sadly, a foodborne illness outbreak happened due to unsanitary conditions. The company learned its lesson and implemented strict SSOPs. Since then, they have kept impeccable cleanliness standards, and earned back their customers’ trust.
Are you ready to join GMPs and follow the rules like a pro?
GMP’s – Good Manufacturing Practices
GMP’s, or Good Manufacturing Practices, are essential for companies to ensure the quality and safety of their products. These practices cover things like facility cleanliness, employee training, and product labeling.
Let’s take a look at the importance of GMP’s. Here’s a table with some key elements:
| Element | Description |
|---|---|
| Facility Design | Having the right infrastructure for safe production |
| Equipment Calibration | Regular checks to keep accuracy and reliability |
| Raw Material Control | Strictly monitoring the ingredients and sources |
| Quality Control Testing | Testing protocols to make sure product’s quality is okay |
| Documentation Procedures | Detailed records for traceability and accountability |
GMP’s also emphasize proper hygiene practices by employees. This includes washing hands, wearing protective clothing, and regular cleaning of equipment.
A real-life example of GMP’s comes from a pharmaceutical company which didn’t follow them. Due to poor sanitation at their manufacturing facility, contaminated medicine was distributed in the market. This led to health issues among consumers, as well as financial losses for the company.
This shows us how following GMP’s is important not only for product quality, but also for consumer safety and protecting a company’s reputation.
Comparing PRP’s, CCP’s, SSOP, and GMP’s
Let’s unravel the key differences between PRP’s, CCP’s, SSOP, and GMP’s! Here’s a comparison table:
| PRP | CCP | SSOP | GMP | |
|---|---|---|---|---|
| Definition | Prerequisite Program | Critical Control Point | Sanitation Standard Operating Procedure | Good Manufacturing Practice |
| Purpose | Preventive measures | Focuses on specific hazards | Comprehensive cleaning | Ensures consistent quality of products |
| Implementation | Entire process | Critical points | On-site sanitation | All manufacturing operations |
| Importance | Food safety foundation | Identifies and controls risks | Maintains cleanliness | Regulatory compliance & customer trust |
PRP’s are a vital base for food safety, setting up preventive steps all through the production process. CCP’s target particular dangers and are applied at critical steps to recognize and regulate risks properly. SSOP guarantees comprehensive cleaning with on-site sanitation procedures. GMP, meanwhile, ensures consistent product quality by observing rigorous manufacturing practices across all operations.
Pro Tip: Regularly review and update these programs to keep up with changing regulatory requirements and industry standards. Now you know what those acronyms mean – PRP’s, CCP’s, SSOP, and GMP’s!
Difference Between PRPs CCPs SSOP and GMPs
PRP’s, CCP’s, SSOPs, and GMPs it’s time to get educated! Each plays a crucial role in food safety and quality control. Understanding the differences between them is key for achieving high standards.
First up are PRP’s (Prerequisite Programs). They form the basis of food safety management systems. They focus on hygiene practices, training, pest control, sanitation, and supplier selection. Implementing effective PRP’s is vital to prevent food hazards.
Next, CCP’s (Critical Control Points) are steps in the production process where control can reduce hazards. Monitoring these points is crucial for minimizing food safety risks. Identifying CCP’s allows targeted interventions in the production process.
SSOP (Sanitation Standard Operating Procedures) provides instructions for cleaning and sanitizing equipment and facilities. It outlines procedures to maintain a hygienic environment throughout production. Implementing SSOP correctly helps prevent cross-contamination and product safety.
Lastly, GMP’s (Good Manufacturing Practices) are guidelines that regulate operations in food processing facilities. These cover personnel hygiene, facility maintenance, equipment calibration, raw material handling, and labeling requirements. Following GMP’s ensures consistency in quality and safety during manufacturing.
Don’t stay behind, update your knowledge on these programs to ensure you’re not missing out! Excellence in food safety is a commitment that we must all embrace.
Frequently Asked Questions

1. What is a PRP?
Prerequisite Programs or PRPs, are a collection of guidelines and methods for establishing sanitary conditions. Examples include pest control and employee hygiene training.
2. What does CCP stand for?
Critical Control Points are referred to as CCP. These are certain production phases where risks can be reduced or avoided. These manufacturing process processes limit risks to a tolerable level.
3. What is SSOP?
SSOP, or Sanitation Standard Operating Procedures, refers to a set of protocols and guidelines that outline the necessary steps for maintaining cleanliness and ensuring food safety in food processing facilities. SSOPs are critical for preventing contamination and adhering to food safety regulations.
4. What does GMP stand for?
GMP stands for Good Manufacturing Practices. It is a set of quality control guidelines and standards that ensure the consistency, safety, and quality of products in industries such as pharmaceuticals, food processing, and cosmetics.
5. How are PRPs and CCPs related?
PRPs and CCPs are directly related. Prerequisite Programs or PRPs are preventive measures that use Critical Control Points that maintain cleanliness and ensure food safety.
6. Are SSOPs and GMPs interchangeable?
No, SSOPs and GMPs are not interchangeable. While both focus on quality and safety, SSOPs specifically address sanitation and cleanliness protocols in food processing facilities, while GMPs encompass broader quality control practices applicable to various industries.
















Leave a Reply