FDA
Numerous compliance standards across various industries require organizations to establish and maintain comprehensive policies and procedures. The specific standards applicable to a business depend on its industry, geographic location, and the nature of its operations. what compliance standards require policies and procedures?
Read moreISO is an international organization that sets standards to make sure products and services are safe, reliable, and consistent. It offers certification to show a company’s commitment to quality assurance, giving it a competitive edge. What’s the difference between ISO vs. FDA?
Read moreSOP, GMP, and procedures: three vital components for efficiency and compliance in various industries. SOPs provide step-by-step instructions for tasks.
Read moreThe FDA plays a crucial role in ensuring medical product safety and efficacy. Its regulatory framework consists of numerous rules that guide industry practices. These are versatile and cater to different product categories. To comply, businesses must understand the relevant requirements.
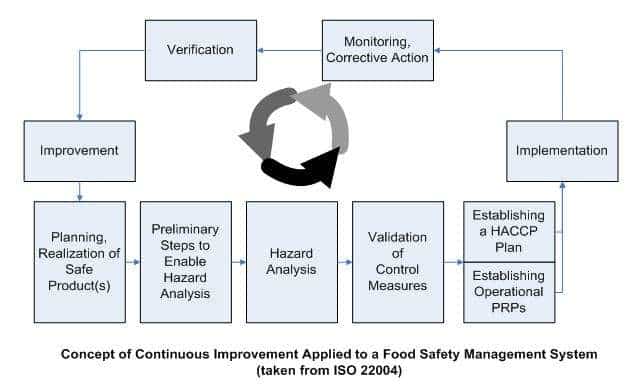
Read moreLet’s start off with a blast! PRP’s, CCP’s, SSOP, and GMP’s – it may seem like alphabet soup at first. But don’t worry, we’ll unravel it all! It’s clear these acronyms play and important part in keeping consumers safe. The FDA even requires companies to use science-driven risk assessments to prevent food safety issues. So remember: […]
Read moreIn today’s fast-paced world, work instructions are key for efficient and effective workplaces. These standards are usually set by industry experts or regulatory bodies and form the basis of operations. Who Sets Work Instruction Standards? Various entities. Professional organizations specialize in a certain field or industry. They bring together experts to create best practices and standards.
Read more