FDA Inspections Procedure Template Word
The FDA Inspections Procedure Template Word is a comprehensive document that outlines the procedures and guidelines for conducting FDA inspections. This template is designed to help businesses prepare for and successfully navigate FDA inspections, ensuring compliance with all relevant regulations and guidelines.
The template includes detailed instructions on how to prepare for an FDA inspection, including how to gather and organize all necessary documentation and how to train employees on FDA regulations and guidelines. It also provides guidance on how to conduct the actual inspection, including how to interact with FDA inspectors and how to respond to any findings or observations.
With the FDA Inspections Procedure Template Word, businesses can ensure that they are fully prepared for FDA inspections and can minimize the risk of non-compliance. The template is easy to use and can be customized to meet the specific needs of any business, regardless of size or industry.
Key features of the FDA Inspections Procedure Template Word include:
- Step-by-step instructions for preparing for and conducting FDA inspections
- Guidance on how to interact with FDA inspectors and respond to findings or observations
- Customizable to meet the specific needs of any business
- Comprehensive and easy to use
Overall, the FDA Inspections Procedure Template Word is an essential tool for any business that is subject to FDA inspections. By using this template, businesses can ensure that they are fully prepared for inspections and can maintain compliance with all relevant regulations and guidelines.
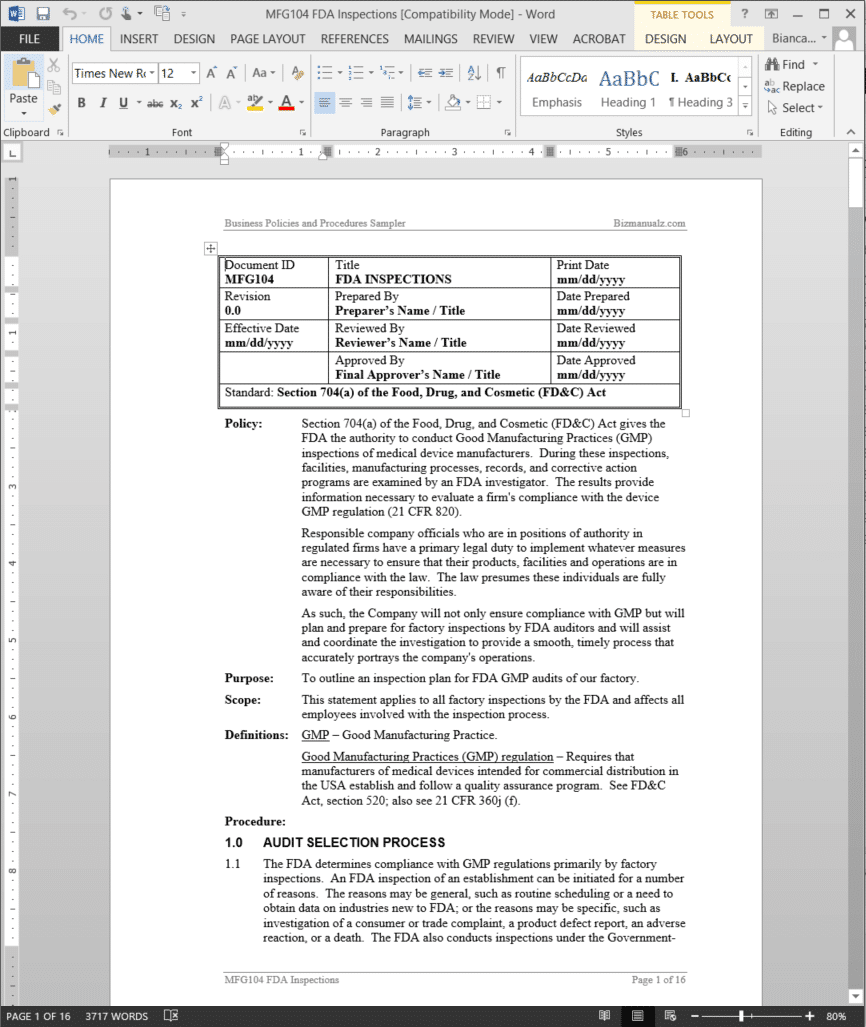
FDA Inspections Procedure
Responsible company officials who are in positions of authority in regulated firms have a primary legal duty to implement whatever measures are necessary to ensure that their products, facilities and operations are in compliance with the law. The law presumes these individuals are fully aware of their responsibilities.
As such, your company will not only ensure compliance with GMP but will plan and prepare for factory inspections by FDA auditors and will assist and coordinate the investigation to provide a smooth, timely process that accurately portrays your company’s operations.
The FDA Inspections Procedure outlines an inspection plan for FDA GMP audits of our factory.This statement applies to all factory inspections by the FDA and affects all employees involved with the inspection process. (16 pages, 3726 words)
FDA Inspections Definitions:
GMP – Good Manufacturing Practice.
Good Manufacturing Practices (GMP) regulation – Requires that manufacturers of medical devices intended for commercial distribution in the USA establish and follow a quality assurance program. See FD&C Act, section 520; also see 21 CFR 360j (f).
 FDA Inspections Procedure Activities
FDA Inspections Procedure Activities
- Audit Selection Process
- Inspection Announcement Protocol
- Conduct During the Inspection
- Areas of Inspection
- Close-Out Meeting and Post-Inspection
FDA Inspections Procedure References
- State and Regulatory Requirements
FDA Inspections Procedure Forms
- Triggers & Decision Points for GMP Inspections
- Excerpts of the FDA’s Two Track GMP Inspection Strategy and Compliance Program






















Reviews
There are no reviews yet.