What Are The Top Quality Manager Responsibilities?

As the Quality Manager you are responsible for Quality Management System (QMS) compliance and strategies to improve quality management. In other words, you must manage all company-wide, quality policies, procedures, processes, programs, and practices, to assure the company of continuous conformance with appropriate standards and regulations. What are the top Quality Manager responsibilities?
Top Ten Quality Manager Responsibilities

The Quality Manage is responsible for overall development, implementation, and maintenance of the organization’s Quality Management System (QMS).
In a smaller company you may also be the document control manager, quality auditor, and process improvement specialist. Listed here are the top Quality Manager responsibilities:
1. Quality Standards
Documents and records are used all throughout the company. Nobody should know more about quality, compliance and processes than the Quality Manager. Your experience with Good Manufacturing Practices (GMP), ISO standards (i.e. ISO 9001), Sarbanes Oxley, or 21 CFR 820 within a regulated industry are important to managing and controlling documents and records.
2. Quality Project Management
A Quality Manager is also a project manager managing corrective action, process improvement, and auditing projects. One must have strong self-motivation, the ability to work independently, and within a team environment with strong follow up, organization and prioritization skills and excellent attention to detail helps too. Perhaps most important of all is understanding the financial ” risk-reward ” trade-offs in good project management.
3. Business Process Documentation
A Quality Manager must be able to understand, comply, and improve established company policies and procedures. Developing standard work, policies, procedures, job aids, and business process communications are a part of the job. A Quality Manager is also a technical writer. Familiarity with policy and procedure writing will help you to succeed. Technical writing conveys technical information using active voice construction, instructional design, and desktop publishing methods to transfer information into understandable and useful information.
4. Document Control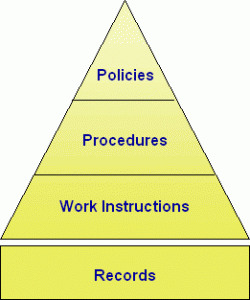
As the Quality Manager, your Knowledge of the Quality documentation process, Document Control practices, and managing documents, records, forms, and work instructions is vital to maintaining your company’s compliance program. A Quality Manager may be the Document Control Manager responsible for organizing documents into an easy to use and fast retrieval system.
Users need their policies and procedures to conform to requirements. If they cannot find them, then they cannot follow them — Document control is an important priority.
5. Quality Communications
It might go without saying but explaining business process compliance to others is what a Quality Manager has to do. Being able to understand a variety of instructions furnished in written, oral, diagram, or schedule form helps others to follow and conform to the established best practice.
Communicating compliance and conformance is done using software such as Microsoft Word and Excel, training using PowerPoint, process mapping using Visio, Statistical Process Control (SPC) using statistics, as well as document revision control software for policies and procedures management.
6. People Management
A Quality Manager should not be afraid of asking questions, collecting business process information, and working with others in a positive and collaborative manner. Business processes include sensitive accounting and financial processes too. So, you must be comfortable speaking effectively and communicating directly with all levels of personnel.
7. Quality Auditing
A Quality Manager performs some of the quality audits. Supply chain audits, process audits, and may even lead ISO audits an act as a Lead Auditor managing teams of auditors. Clearly understanding the compliance requirements, collecting objective evidence, and writing up audit reports ensure the quality management system is operating effectively.
8. Problem Solving
The Quality Manager solves problems, typically in situations where general standardization should exist, but may not be operating effectively. Using process mapping techniques, lean, or Six Sigma process improvement methodology is essential to reducing waste and being effective in the Quality role.
A delicate balance exists between resolving problems yourself and identifying those situations that require management intervention for a solution. Good political judgment is required where quality and compliance are concerned.
9. Team Player
The Quality Manager is involved in team building and meetings at every level of the organization. Management reviews, material reviews, supplier reviews, corrective action reviews, process improvement teams, audits, customer visits and strategy discussions. A good Quality Manager is involved with many areas of the company.
10. Quality “Go To” Guy
The Quality Manager is the person that everybody goes to to get answers about quality. Product specifications, supplier requirements, testing, inspections, part verification, equipment calibration, corrective actions, non-conformances, workers compensation, benchmarking, voice of the customer, and on and on. The buck stops here at Quality.
Top Ten Quality Manager Responsibilities
As the Quality Manager you are responsible for Quality Management System (QMS) and all of its related compliance. You manage quality policies, procedures, processes, programs, and practices, to assure the company of continuous improvement, conformance and effectiveness. In a smaller company you may also be the document control manager, quality auditor, and process improvement specialist. But you are always a team player that has input on anything to do with quality.
















Leave a Reply