ISO Compliance Procedure Template Word
The ISO Compliance Procedure Template Word is a comprehensive document that helps organizations comply with the International Organization for Standardization (ISO) standards. This template is designed to assist businesses in creating a systematic approach to meet the requirements of ISO standards, which are essential for maintaining quality, safety, and environmental management systems.
The ISO Compliance Procedure Template Word is easy to use and customizable, allowing businesses to tailor the document to their specific needs. The template includes step-by-step instructions, sample procedures, and forms that can be easily modified to meet the requirements of any ISO standard. The document is available in Microsoft Word format, making it easy to edit and share with team members.
By using the ISO Compliance Procedure Template Word, businesses can ensure that they are meeting the requirements of ISO standards, which can help them improve their operations, reduce risks, and increase customer satisfaction. The template covers a wide range of ISO standards, including ISO 9001, ISO 14001, ISO 45001, and more.
Overall, the ISO Compliance Procedure Template Word is an essential tool for any business that wants to comply with ISO standards. It provides a comprehensive approach to meeting the requirements of ISO standards, which can help businesses improve their operations, reduce risks, and increase customer satisfaction. With its easy-to-use format and customizable features, the ISO Compliance Procedure Template Word is a must-have for any organization that wants to achieve ISO compliance.
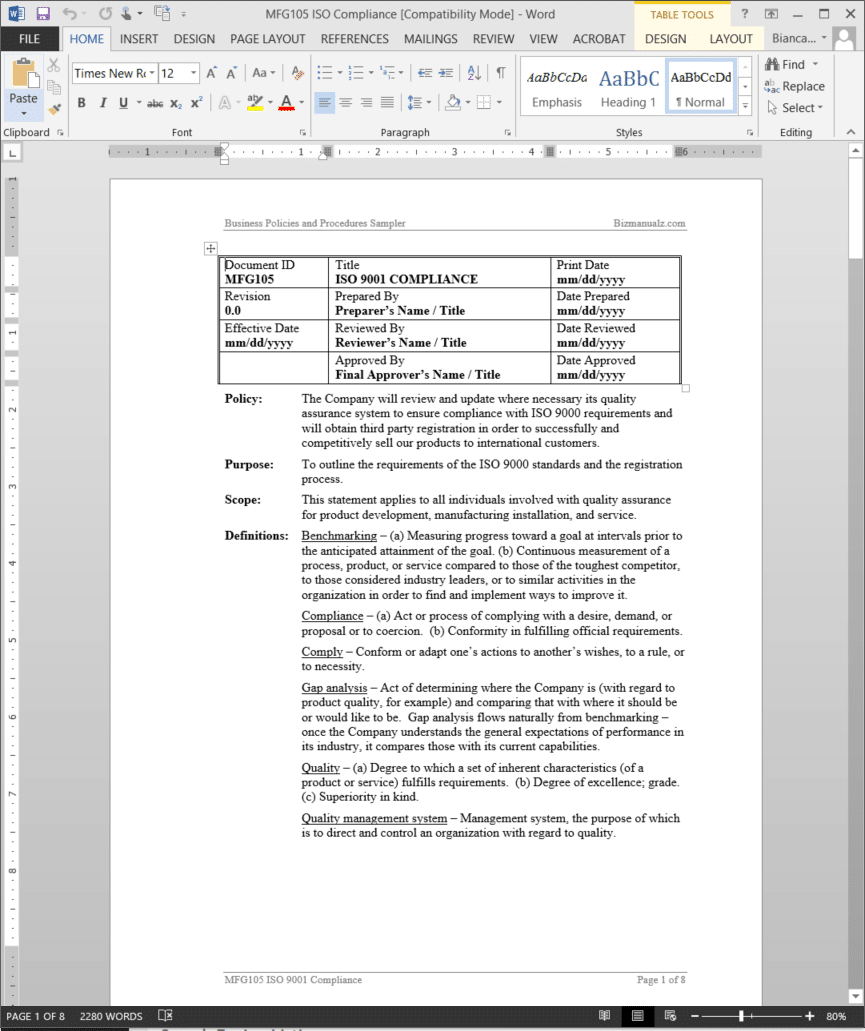
ISO Compliance Procedure
It’s important for your company to review and update where necessary its quality assurance system to ensure compliance with ISO 9001 requirements and will obtain third party registration in order to successfully and competitively sell our products to international customers.
The ISO Compliance Procedure outlines the requirements of the ISO 9001 standards and the registration process. This statement applies to all individuals involved with quality assurance for product development, manufacturing installation, and service. (8 pages, 2315 words)
ISO Compliance Definitions:
Benchmarking – (a) Measuring progress toward a goal at intervals prior to the anticipated attainment of the goal. (b) Continuous measurement of a process, product, or service compared to those of the toughest competitor, to those considered industry leaders, or to similar activities in the organization in order to find and implement ways to improve it.
Compliance – (a) Act or process of complying with a desire, demand, or proposal or to coercion. (b) Conformity in fulfilling official requirements.
Comply – Conform or adapt one’s actions to another’s wishes, to a rule, or to necessity.
Gap analysis – Act of determining where the company is (with regard to product quality, for example) and comparing that with where it should be or would like to be. Gap analysis flows naturally from benchmarking – once the company understands the general expectations of performance in its industry, it compares those with its current capabilities.
Quality – (a) Degree to which a set of inherent characteristics (of a product or service) fulfills requirements. (b) Degree of excellence; grade. (c) Superiority in kind.
ISO Compliance Procedure Activities
- ISO Background and ISO Standards
- Overview of ISO 9001
- Quality System Objectives and Principles
- Quality Management System Development
- ISO Registration
ISO Compliance Procedure References
- ISO/EN 9001:2008, Quality Management Systems ” Requirements, 4th edition, Dec. 15, 2008






















Reviews
There are no reviews yet.